Vision | Mission | Need for the Centre | Leadership | Members
Vision
The CPO aims to develop molecular diagnostics that will improve the detection of cancer and will characterize tumours for selection of the most effective molecularly-targeted precision cancer treatments. The CPO also aims to develop more effective treatments for cancers that have poor prognosis for which there are currently few treatments.
Mission
The CPO aims to bridge the gap between research discoveries in cancer biology and the development of new molecular diagnostics and therapeutics for cancer and their advancement to first-in-humans clinical trials. The CPO has an educational mandate to train the pharmaceutical oncology scientists of the future through offering courses and research training.
Need
There is a compelling need to develop new and improved diagnostic tools and treatments for cancer, particularly for cancer in its advanced stages and for cancers for which there are currently no adequate treatments. The Leslie Dan Faculty of Pharmacy at the University of Toronto, as Canada’s leading Faculty of Pharmacy with the largest graduate Department of Pharmaceutical Sciences with more than 150 students pursuing MSc and PhD degrees is uniquely positioned to take the lead in pharmaceutical oncology. The location of the Faculty in the heart of the Toronto health sciences research community which has world-leading strengths in cancer biology, molecular diagnostics and cancer treatment including extensive clinical trials infrastructure provides the CPO with the expert collaborations and translational resources needed to succeed in this challenge.
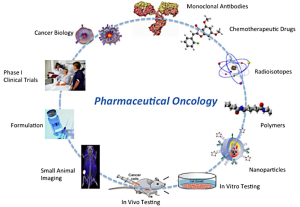
Basic discoveries in cancer biology and clinical trials infrastructure are the two pillars needed to make transformative change in the outcome of cancer patients. The Centre for Pharmaceutical Oncology aims to bridge these two pillars by developing the molecular diagnostics and therapeutics for cancer that build on a better understanding of cancer biology and advance the most promising agents to clinical trials.
Discoveries in cancer biology provide opportunities for the development of new diagnostics and therapeutics for cancer that include monoclonal antibodies or small molecule chemotherapeutic drugs. These may be linked to radioisotopes for imaging or treatment or to polymers to improve their pharmaceutical properties. These agents may be incorporated into advanced drug delivery systems (e.g. nanoparticles) or monoclonal antibodies may be used to target these systems to tumours. Following in vitro and in vivo studies including state-of-the-art small animal imaging studies, these new diagnostics and therapeutics may be formulated in pharmaceutical quality for Phase I/II clinical trials in cancer patients. The results of these trials will inform on the design of next generation agents, completing the innovation cycle.
Leadership
 Dr. Raymond Reilly– Director and CPO executive committee member – Profile
Dr. Raymond Reilly– Director and CPO executive committee member – Profile
 Dr. Christine Allen – CPO executive committee member – Profile
Dr. Christine Allen – CPO executive committee member – Profile
 Dr. Stephane Angers – CPO executive committee member – Profile
Dr. Stephane Angers – CPO executive committee member – Profile
Members
List of CPO Members coming soon.